Prof. Peng, I-Chen

中文
TEL:+886-6-275-7575 ext.58123 研究室:58104 or 58114轉61
Email: ipeng@mail.ncku.edu.tw
Web:Laboratory Website
Research interests:
Molecular Medicine, Nutrition and Epigenetics, Cancer Metabolism
Introduction of Laboratory:
Dr. Peng’s research focuses on targeting lipid and glutamine metabolism to treat obesity and obesity-related diseases, particularly the molecular mechanisms of oncogenic signaling and epigenetic regulation of lipid and glutamine metabolism in adipocytes, hepatocytes and cancer cells. The long term goal is to determine the key enzymes for targeting in cancer metabolism and translation to the clinic.
Research Summary:
Alteration of lipid metabolism has been increasingly recognized as a hallmark of obesity andobesity-related diseases, i.e. cancer. Oncogenic growth signaling upregulates glycolysis, lipogenesis and glutamine metabolism to meet the biosynthetic demands of rapidly proliferating tumor cells. However, how cancer cells manipulate glucose, lipid and glutamine metabolism is unclear. The metabolic sensor AMPK is stimulated upon fasting, exercise and calorie restriction to augment lipid and glucose metabolism. Oncogenic Myc has been shown to favor the conversion of glucose to lactate and increase the usage of glutamine in cancer cells. Given that metabolic reprogramming is modulated by circadian-, nutritional-, and environmental-driven molecular signals, epigenetic alterations of metabolic gene expression are dynamic and reversible. Recently, we have discovered that Myc functions as an epigenetic regulator to increase the expression of glutamine synthetase. Therefore, we are interested in investigating mechanotransduction (i.e., AMPK and Myc signaling), transcriptional and epigenetic regulation of lipogenesis and glutamine metabolismin in adipocytes, hepatocytes and cancer cells. Our long term goal is to determine the key enzymes for targeting in cancer metabolism and translation to the clinic.
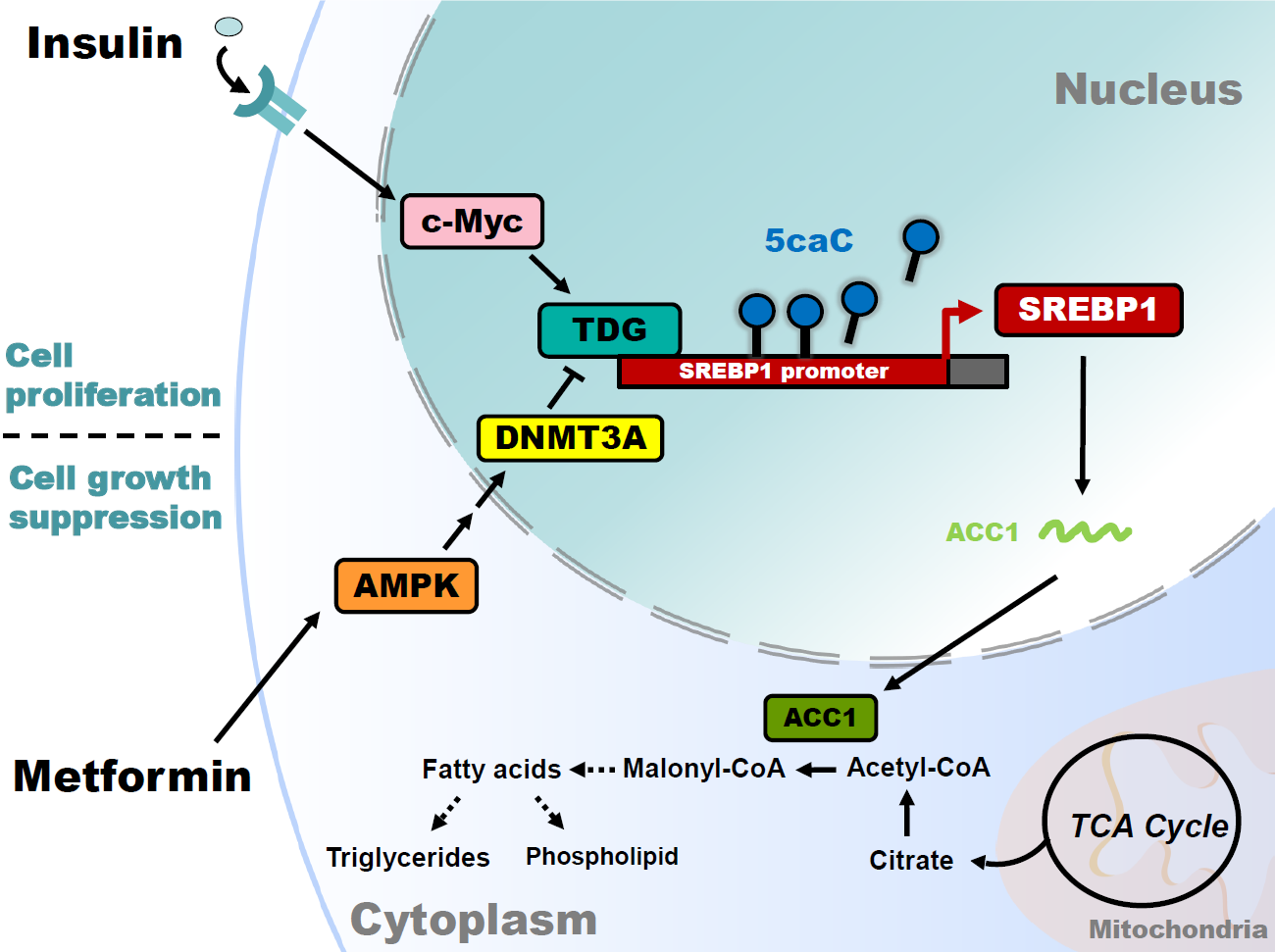
Current research projects include:
- Study the molecular link between insulin and metformin signaling with cancer metabolism.
- Investigate the epigenetic regulation of lipid and glutamine metabolism in adipocytes, hepatocytes and cancer cells.
- Reveal the biological impacts of lipid droplets on cancer proliferation and drug resistance.
Education
| Ph.D. Degree | Graduate Program in Biochemistry and Molecular Biology, University of California, Riverside, CA, USA |
| Master Degree | Graduate Institute of Agricultural Chemistry, Microbiology Division, National Taiwan University, Taiwan |
| Bachelor’s Degree | Department of Agricultural Chemistry, National Taiwan University, Taiwan |
Experience
| 2020 Associate Professor | Department of Life Sciences, National Cheng Kung University, Taiwan |
| 2014 Assistant Professor | Department of Life Sciences, National Cheng Kung University, Taiwan |
| 2012 Postdoctoral Fellow | Department of Molecular Genetics and Microbiology, Stony Brook University, NY, USA |
| 2011 Postdoctoral Fellow | Division of Biomedical Sciences, University of California, Riverside, CA, USA |
Awards and Honors
Publications
-
- Jin-Wei Jhu, Jia-Bao Yan, Zou-Han Lin, Shih-Chieh Lin, and I-Chen Peng*. (2021 Sep). SREBP1-induced glutamine synthetase triggers a feedforward loop to upregulate SREBP1 through Sp1 O-GlcNAcylation and augments lipid droplet formation in cancer cells. International Journal of Molecular Sciences.22(18), 9814. (* for correspondence)
- Jia-Bao Yan, Chien-Cheng Lai, Jin-Wei Jhu,Brendan Gongol, Traci L. Marin, Shih-Chieh Lin, Hsiang-Yi Chiu, Chia-Jui Yen, Liang-Yi Wang, and I-Chen Peng*. (2020 Jun) Insulin and metformin control cell proliferation by regulating TDG-mediated DNA demethylation in liver and breast cancer cells. Molecular Therapy-Oncolytics. 18: 282-294. (* for correspondence)
- Pai-Sheng Chen, Wen-Tai Chiu, Pei-Ling Hsu, Shih-Chieh Lin, I-Chen Peng, Chia-Yih Wang , and Shaw-Jenq Tsai. (2020 May) Pathophysiological implications of hypoxia in human diseases. Journal of Biomedical Science. 27(1):63.
- I-Chen Peng*, Alex J. Bott, and Wei-Xing Zong*. (2016 Oct) Spectrophotometric determination of glutamine synthetase activity in cultured cells. Bio-protocol 6(19): e1959. (* for correspondence)
- Alex J. Bott*, I-Chen Peng* (co-first author), Yongjun Fan, Brandon Faubert, Lu Zhao, Jinyu Li, Sarah Neidler, Yu Sun, Nadia Jaber, Dawid Krokowski, Wenyun Lu, Ji-An Pan, Scott Powers, Joshua Rabinowitz, Maria Hatzoglou, Daniel J. Murphy, Russell Jones, Song Wu, Geoffrey Girnun, and Wei-Xing Zong. (2015 Dec) Oncogenic Myc induces expression of glutamine synthetase through promoter demethylation. Cell Metabolism. 22(6):1068-1077. (* contributed equally to this study)
- Yu-Ting Hung, Chi-Te Liu, I-Chen Peng, Chin Hsu, Roch-Chui Yu, Kuan-Chen Cheng. (2015 Sep) The implementation of a Hazard Analysis and Critical Control Point management system in a peanut butter ice cream plant. Journal of Food and Drug Analysis. 23(3):509-515.
- Brendan Gongol, Traci Marin, I-Chen Peng, Brian Woo, Stephanie King, Marcy Martin, Wei Sun, Shu Chien, and John Y-J. Shyy. (2013). AMPKa2 exerts its anti-inflammatory effects through PARP-1 and Bcl-6. Proc Natl Acad Sci U S A. 110(8):3161-3166.
- I-Chen Peng, Kai-Yin Lo, Chun-Hua Hsu, and Chia-Yin Lee. (2012). Increasing the storage and oxidation stabilities of N-acyl-D-amino acid amidohydrolase by site-directed mutagenesis of critical methionine residues. Process Biochemistry. 47:1785-1790.
- I-Chen Peng, Zhen Chen, Wei Sun, Ying-Shiuan Li, Traci LaNai Marin, Pang-Hung Hsu, Mei-I Su, Xiaopei Cui, Songqin Pan, Christian Y. Lytle, David A. Johnson, Frank Blaeser, Talal Chatila, and John Y-J. Shyy. (2012). Glucagon regulates ACC activity in adipocytes through the CaMKKb/AMPK pathway. Am J Physiol Endocrinol Metab. 302(12):E1560-1568.
- Zhen Chen, I-Chen Peng (co-first author), Xiaopei Cui, Yi-Shuan Li, Shu Chien, and John Y-J. Shyy. (2010). Shear stress, SIRT1, and vascular homeostasis. Proc Natl Acad Sci U S A. 107(22):10268-10273.
- Zhen Chen, I-Chen Peng, Wei Sun, Mei-I Su, Pang-Hung Hsu, Yi Fu, Yi Zhu, Kathryn DeFea, Songqin Pan, Ming-Daw Tsai, and John Y-J. Shyy. (2009). AMP-activated protein kinase functionally phosphorylates endothelial nitric oxide synthase Ser633. Circ Res. 104:496-505.









